Quorum sensing inhibition in animal nutrition
As an ETEC, you have a special power: by secreting certain heat-stable enterotoxins, you are able to form pores in the jejunal brush border membrane, leading to diminished absorptive ability of the villi and secretion of water and electrolytes.
As an ETEC, you have a special power:
by secreting certain heat-stable enterotoxins, you can form pores in the jejunal brush border membrane, leading to diminished absorptive ability of the villi and to secretion of water and electrolytes. Thus, you have the power to start intestinal diseases like post-weaning diarrhea (PWD) in piglets. As many other bacterial species, you use your flagellum for moving around and your fimbriae to attach firmly to the receptors of the gut epithelial cells. You like your new environment. The microflora in the intestines that can stop you is still very immature, and you can grow faster than most of them.
But at this moment you are relatively harmless, as there are only a few family members simultaneously attaching to the inner wall of the intestine. Because you belong to the rather “communicative type” of bacteria, you are constantly secreting signal molecules to your environment, known as autoinducers (AIs). Being gram-negative, you use N-acyl-homoserine lactones (AHLs) as AI, whose production depends on S-adenosylmethionine as a substrate. Since these AIs have amphiphilic properties, they can pass through the cell membrane of other E. coli colleagues in your surroundings easily.
Since the density of your E. coli group is still very low…
…there is only a low AIs concentration in your residential milieu. But not for long: The inflowing intestinal content provides enough nutrients to you and your family and you have optimum conditions to start multiplying yourself via cell division. As the number of E. coli around you increases and they are all like you of the ‘communicative type’, more and more AIs are secreted. Due to the increased density of bacteria and AIs, the likelihood of a secreted AI to diffuse into another bacterium in the immediate vicinity is enhanced. This creates a strong network of AIs and powerful communication among each other – known as Quorum Sensing (QS).
What’s the purpose of Quorum sensing?
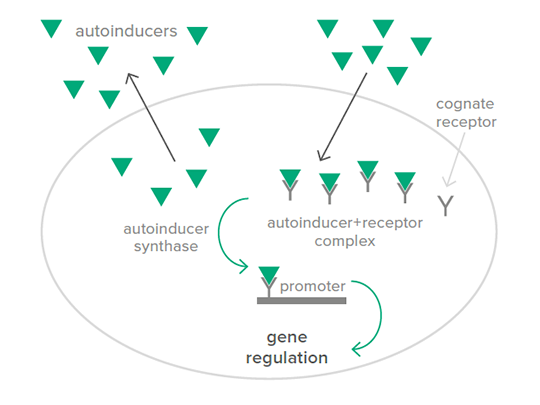
And what happens with those AIs, diffusing into the bacterial cell? The AIs bind to receptors, from which AI-receptor complexes arise, acting simultaneously as transcription factors. Thus, with binding to the receptors, the expression of genes in the QS regulon is regulated. Subsequently, a positive feedback loop is generated: With an increasing concentration of AIs, the transcription of specific genes is activated, including those involved in the production of additional AIs as well as of toxins or adhesion factors like fimbriae.
As stated above, only if a certain population-density of you and your family members is reached, the synthesis of virulence factors like enterotoxins will be triggered. These enterotoxins increase the permeability of the intestinal wall of your host, resulting in lower nutrient absorption, as well as the leak of water and electrolytes into the intestine. Consequently, post weaning diarrhoea (PWD) sets in: the body of your host loses nutrients and water– it literally dries out. If left untreated, this condition quickly causes death.
With the use of QS…
…pathogenic bacteria similarly behave like a complex organism, making „collaborative decisions”, such as expressing genes for producing enterotoxins, forming biofilms, develop genetic competence and much more. Moreover, QS seems to help you to measure the size of the population around you and to adapt their behavior. Thus, QS gives you several advantages: becoming stronger than most other bacterial strains ensures protection and survival of your group.
Suddenly…
… it gets quiet: the communication stops, you don’t hear anyting. What’s going on? Someone seems to have fed the piglet some phytogenic feed additives (PFAs). Some plant-based bio-actives have the ability to “interrupt” the communication with your family, due to so-called quorum sensing inhibition (QSI). This means, even if a specific cell density is exceeded, no virulence factors are secreted, and the bacterial community stays harmless to the host organism.

Quorum sensing inhibition in animal nutrition?
Quorum sensing inhibition in animal nutrition is a valuable tool. Let’s have a look on the beneficial quorum sensing inhibitory effects of selected phytogenics: Polyphenols for example show the ability to inhibit quorum sensing and thus affecting biofilm formation in some bacteria. As they do not affect the intestinal microbiota, they prevent simultaneously the concurrent invasion of pathogens by supporting a balanced microbiota. Other components like curcumin, the active substance of Curcuma longa root, inhibits the expression of virulence genes in Pseudomonas aeruginosa.
But how is Quorum sensing inhibition working?
There exist quite numerous mechanisms, as to how phytogenics interrupt QS in gram-negative bacteria. Several plant extracts, like phenolics, flavonoids, alkaloids and terpenoids may act as Quorum sensing inhibitiors via different mechanisms, such as inhibition of AHL synthesis, transport or secretion, binding and storage of AHLs, antagonistic actions and inhibition of targets downstream of AHL receptor binding.
Let’s have a closer look on them:

Furanones, contained in strawberries for instance, interfere with AI-regulated processes in Pseudomonas aeruginosa by accelerating the degradation of the AI-receptor protein. Moreover, isolimonic acid, an active compound of citrus fruits-seeds interferes biofilm formation in E. coli due to inhibition of AI-activity.
As a consequence, attachment of the AI molecule to the target receptor, initiating gene expression, does not take place. Thus, synthesis of e.g. virulence genes and biofilms remain without affecting cell viability. Moreover, carvacrol, one of the antimicrobial components in oregano oil as well as of peppermint interferes with AHL regulated virulence factors and thus inhibits biofilm formation of several gram-negatives.

Piperine, an ingredient of black pepper, exhibits its quorum sensing inhibitory effect by impeding the activity of receptors and molecules involved in the QS pathway, required for biofilm formation of Streptococcus mutans. Naturally occurring furocoumarins from grapefruit unrolled inhibitory effects on autoinducer activities, subsequent biofilm formation of several gram-negative bacteria like E. coli and Salmonella typhimurium.

Moreover, it could be shown that an extract of Mangifera indica, rich in hydrolysed tannins, exhibits a broad spectrum of anti-QS activities including prevention of AHLs activity. The extract had a protein binding activity and thus may disrupt QS either by inactivating enzymes responsible for AIs-synthesis or binding to protein receptors of QS signals.
Due to QS inhibition by plant-based extracts, you as E. coli or other pathogenic bacterium will be no longer able to communicate and to harm the hosts’ organism. That shows that quorum sensing inhibition in animal nutrition can be a valuable tool to support the animals’ resilience and wellbeing.
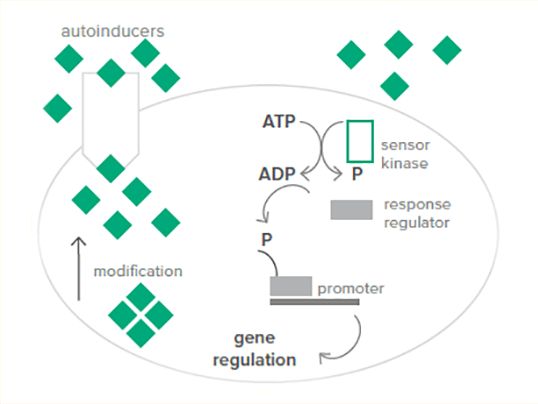
- Gram-positive and Gram-negative bacteria use different types of QS systems
- Gram-positive bacteria use short-peptide-molecules as signaling molecules called autoinducing peptides (AIPs)
- At high extracellular concentration of AIP, occurring on high cell density in gram-positive, the AIPs bind to membrane-bound histidine-kinase receptors
- Those binding activates the receptor’s kinase activity, which leads to a structural change (autophosphorylation) and becomes able to pass phosphate to a cognate cytoplasmic response regulator
- These phosphorylated response regulators now activate gene transcription
Take home messages:
- Enterotoxigenic E. coli and other pathogens communicate via Quorum Sensing (QS)
- QS allows bacteria to align gene regulation in order to form biofilms, toxins etc. only by reaching a certain cell density
- Phytogenics are able to inhibit QS due to different mechanisms

Anne Oberdorf
Anne has always been fascinated by the unknown, the diversity and beauty of nature. Her love for nature brought her to Delacon in 2018 after studying agricultural sciences, where she worked as Technical Communications Manager and later as Product Manager Aquaculture. Since February 2021, she has been taking a new, natural career path outside of Delacon.










