Saponins - less prominent, though not less important plant ingredient - part 1
Autumn is coming and thus, the chestnuts are falling from the trees. Though - what do chestnuts, very popular handicraft utensils, have to do with saponins and why can they be used to clean our laundry? These and some other interesting questions we are going to answer in our current blog article about saponins. Read on and get an overview about a not so well known but not less important plant ingredient.

Essential oils or herbs are natural ingredients, commonly used in daily kitchen work, and a broad audience is familiar with their beneficial effects on flavor or with their impact on the gastrointestinal tract. In contrast to essential oils or herbs, the group of saponins is less well known.
You certainly have already experienced saponins in action in your daily life - be it the foam on beer, in shampoo, toothpaste or simply as soaps. This is due to their surfactant properties, making them common foaming agents in industry. Saponins show diverse favorable biological actions and represent an essential ingredient of Delacon’s phytogenic feed additives. That´s why we´d like to have a closer look at them, including their important beneficial effects on ammonia reduction in farm animals.
Did you know?
Saponins are given their name as they produce a soap-like foam when being shaken with water.

Saponins, from latin sapo = soap, referring to the interfacial activity observed for this class of molecules (1), are a diverse group of natural compounds. They are widely distributed in the plant kingdom, being found in more than 500 higher plant species with most commonly between 0.1-10% saponins present in the plant (2,3,4). Depending on the plant species, saponins can be found in varying concentrations in all tissues such as roots, bulbs, leaves, blossoms, fruit, and bark. Thousands of different saponins are known today, differing in their chemical structure as well as in their biological activity. The high variability can be explained by their complex chemical structure and by the different metabolic synthesis pathways used by plants.

Not less diverse is the list of plants, of which saponins can be extracted from. Next to sources like e.g. soybean, spinach, ginseng, sugar beet, sunflower, chickpea, oats, yes even marine species like starfish, (2,3,6,7,8), main sources of saponins used in medicine and industrial applications are soap bark tree (Quillaja saponaria), Mojave yucca (Yucca schidigera), licorice (Glycyrrhiza species), ginseng (Panax species), fenugreek (Trigonellafoenum‐graceum), alfalfa (Medicago sativa), horse chestnut (Aesculus hippocastanum), soapwort (Saponaria officinaux), gypsophila genus (Gypsophila paniculata) and sarsaparilla (Smilax species) (23).
A brief excursion into the chemistry of saponins
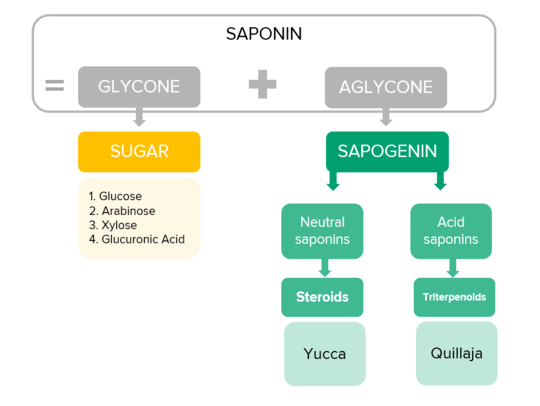
Despite of their high diversity, all saponins consist of the same two molecule groups: a sugar part, called glycone, and a non-sugar part, called aglycone. The aglycone is used to differentiate the saponins, as it is characteristic for this molecule group, and placed at its “core”. Thus, all steroid saponins share a basic aglycone structure, while all triterpenoid saponins have one of two possible characteristic aglycones. The sugars, in contrast, have a high variability and lead to the many different biological functions of the saponins (1,9; see figure 1).
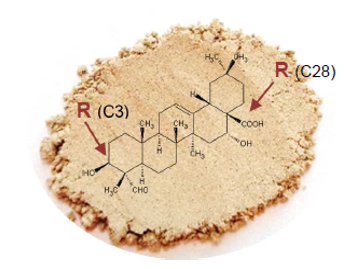
Triterpenoid sapogenins from Q. saponaria consist of a five-ringed quillaic acid backbone (structure see fig 1) with small, frequently branched carbohydrate chains of 2-5 sugar units, attached at the 3‘ and 28‘ carbons of quillaic acid. Main quillaja saponins are: QS-7, -17, -18, -21 (see figure 2).
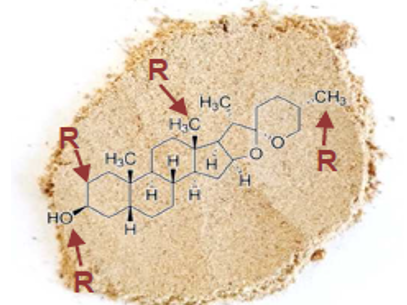
Steroidal sapogenins are predominantly spirostanol saponins (~94%), but also furostanol saponins (~6%). The main spirostanol aglycones are sarsasapogenin and its C-25 epimer smillagenin (66%, structure see fig 2), as well as gloriogenin (24%), Markogenin (3.5%), Tigogenin and Hecogenin (see figure 3).
“Protection shield” for plants
Though in the plant the exact function of many saponins is still unknown, they probably serve as defensive substances, as “shield” for plants, to counter pathogens and herbivores (10) but especially to protect them against fungal attack. Since plants do not have an active immune system like vertebrates, harmful organisms are often combated chemically (5).
Stay tuned - in the next part, we are going to talk about the wide range of applications of saponins, especially focusing on their beneficial effects in emission reduction

Elisabeth Rohrer
After her study in agriculture sciences at the university of natural resources and life sciences in Vienna, Elisabeth joined the Delacon team in December 2013 as Technical Communications Manager - a position, she always exerted with pleasure. Since 2021, her task areas have been extended and thus, she is also supporting colleagues in writing offside the technical focus as Content Manager. Elisabeth describes herself as a great animal and nature lover and prefers to spend her free time high up in the mountains with her little family, away from the hustle and bustle.











I would like to receive the news letter ,information and invitation for Delacon 's activities.
Thank you very much for your feedback.
Very interesting article about the composition of saponins.